FDA Ibuprofen Pregnancy Warning: What You Need to Know
Pregnancy and Medication: A Delicate Balance
When it comes to managing pain and inflammation during pregnancy, many women rely on over-the-counter (OTC) medications like ibuprofen. However, the U.S. Food and Drug Administration (FDA) has issued a warning regarding the use of ibuprofen and other nonsteroidal anti-inflammatory drugs (NSAIDs) during pregnancy. In this article, we will delve into the FDA's warning and provide information on the risks associated with taking ibuprofen during pregnancy.The FDA Warning: A New Approach to Pregnancy Safety
On September 10, 2025, the FDA announced a new warning on the use of NSAIDs during pregnancy. According to the FDA, pregnant women should avoid taking ibuprofen and other NSAIDs for the last four months of pregnancy. This warning is a significant departure from the previous recommendation, which advised against taking these medications during the last three months of pregnancy.Risks Associated with Ibuprofen During Pregnancy
The FDA's warning highlights the potential risks of taking ibuprofen during pregnancy, particularly after 20 weeks. These risks include: * Kidney problems in the unborn baby: The FDA warns that use of NSAIDs around 20 weeks or later in pregnancy may cause rare but serious kidney problems in an unborn baby. This can lead to low levels of amniotic fluid surrounding the baby. * Baby's kidney function: The FDA also warns that NSAIDs may affect the baby's kidney function, which can lead to issues with the baby's kidneys after birth.Alternatives to Ibuprofen During Pregnancy
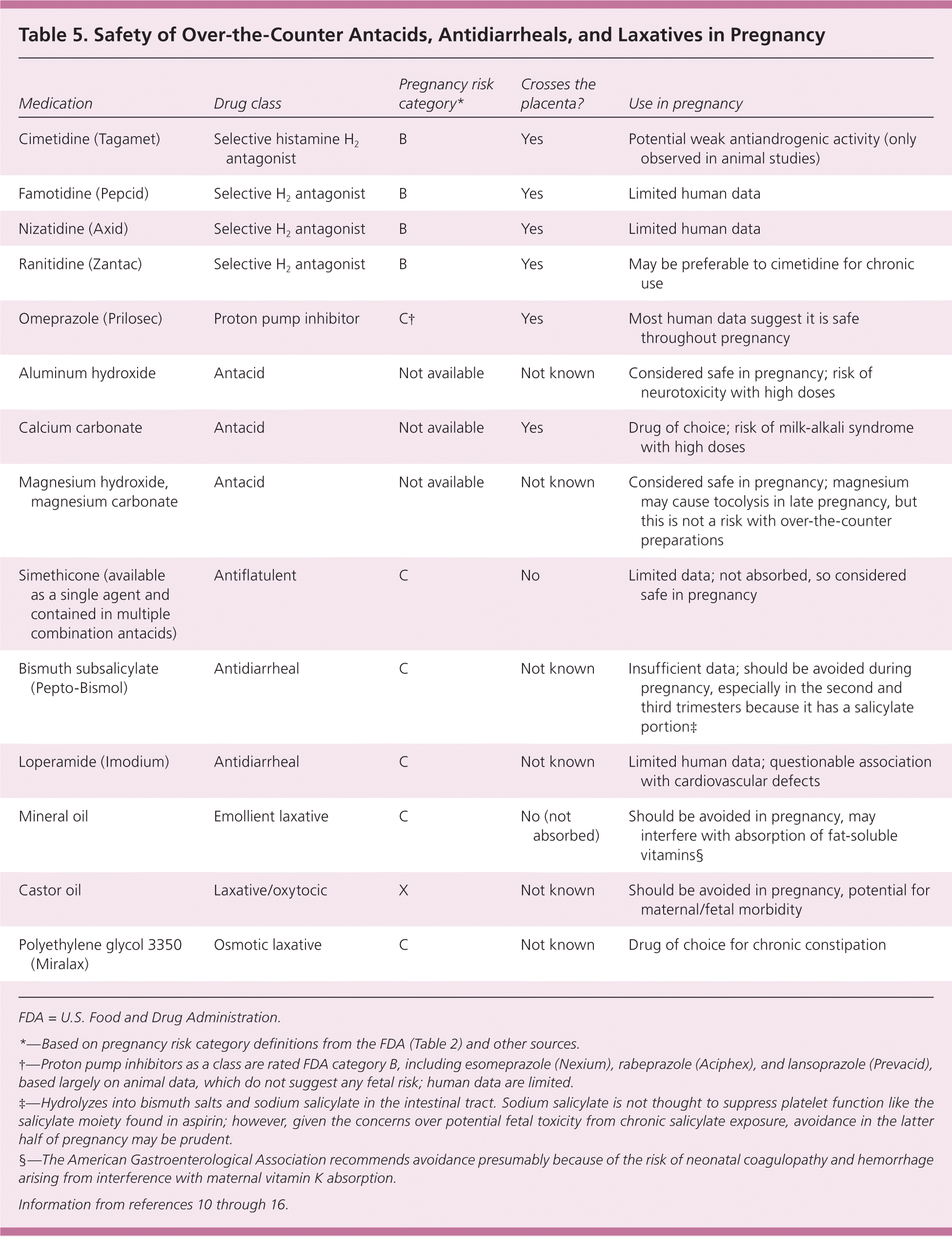
While ibuprofen may not be the best option for pregnant women, there are alternative medications available for managing pain and inflammation. Some of these alternatives include: * Acetaminophen: